Eli Lilly & Co. v. Teva Parenteral Medicines , No. 2011-1561 (Fed. Cir.)
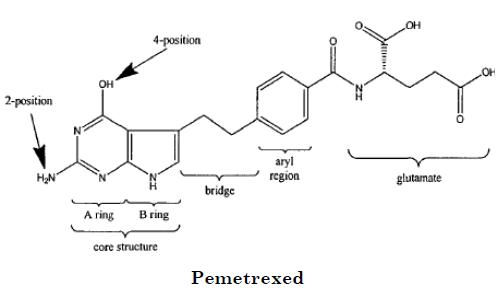
Lilly's anticancer drug Alimta (pemetrexed) is approved for the treatment of mesothelioma and non-small cell lung cancer. Teva, Barr and APP filed ANDAs seeking approval to market generic versions of Alimta prior to the expiration of U.S. Patent No. 5,344,932, owned by Princeton University and exclusively licensed to Lilly.
In a unanimous opinion last Friday, the Federal Circuit affirmed a district court decision that the asserted claims of the '932 patent are not invalid for obviousness-type double patenting.
Claim 3 of the '932 patent recites pemetrexed:
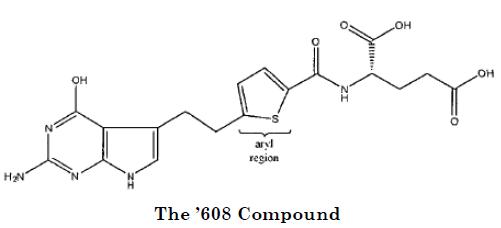
Teva asserted that claim 3 of the '932 patent is invalid for obviousness-type double patenting over two patents in the same family as the '932: U.S. Patent Nos. 5,028,608 and 5,248,775. The '608 and '775 patents have expired, but the '932 patent remains in effect until July 24, 2016, due to a patent term extension of over four years to compensate for delays in the regulatory approval of Alimta. Lilly holds a further six months of pediatric exclusivity over pemetrexed.
As the Federal Circuit explained:
The doctrine of obviousness-type double patenting is intended to prevent the extension of the term of a patent by prohibiting the issuance of claims in a second patent not patentably distinct from the claims of the first patent. A later patent claim is not patentably distinct from an earlier claim if the later claim is obvious over, or anticipated by, the earlier claim.
Teva's first argument was that pemetrexed is not patentably distinct from a compound recited in claim 3 of the '608 patent:
Here, according to the Federal Circuit:
Teva's primary argument concerns the appropriate legal standard for evaluating obviousness-type double patenting. Relying on our decision in Amgen Inc. v. Hoffman La-Roche Ltd., 580 F.3d 1340 (Fed. Cir. 2009), Teva contends that the correct analysis involves only the differences between the claims at issue, so that any features held in common between the claims–in this case, all but the aryl regions of the '608 Compound and pemetrexed–would be excluded from consideration. In Amgen, we explained that once the differences between claims are established, the obviousness-type double patenting analysis entails determining "whether the differences in subject matter between the claims render the claims patentably distinct." But those differences cannot be considered in isolation–the claims must be considered as a whole.
On the merits, the court stated:
In the chemical context, we have held that an analysis of obviousness-type double patenting requires identifying some reason that would have led a chemist to modify the earlier compound to make the later compound with a reasonable expectation of success. Here, the district court considered the parties' arguments and evidence, particularly their conflicting expert testimony as to how an ordinarily skilled chemist presented with the '608 Compound would have been motivated to proceed at the time. In its decision, the court credited Lilly's evidence to find that "the ways in which a person of ordinary skill in the art would modify [the '608 Compound] would not result in pemetrexed." We owe that finding considerable deference on appeal, and we see no clear error based on the record before us.
Teva's second argument was that pemetrexed is not patentably distinct from a compound recited in claim 2 of the '775 patent:
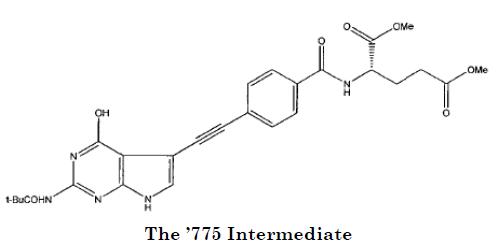
Here, Teva's arguments were based not on the structural similarity of pemetrexed and the '775 Intermediate, but rather on the fact that the '775 patent disclosed that the '775 Intermediate is used to make pemetrexed. As the court put it, "By later claiming pemetrexed itself, Teva maintains, the '932 patent appropriates a previously disclosed use for previously patented compound, which renders the asserted '932 claims invalid for obviousness-type double patenting under a line of our precedent including . . . Sun Pharm. Indus. v. Eli Lilly & Co. (Fed. Cir. 2010)."
The Federal Circuit explained:
As a general rule, obviousness-type double patenting determinations turn on a comparison between a patentee's earlier and later claims, with the earlier patent's written description considered only to the extent necessary to construe its claims. . . . The cases on which Teva relies represent a limited exception to this customary framework. . . . In Pfizer v. Teva Pharms. USA (Fed. Cir. 2008), we held claims to methods of administering a particular anti-inflammatory drug invalid for obviousness-type double pateting where th patentee's earlier patent claimed the drug itself and disclosed the same methods of administering the drug. And in Sun, the patent holder had developed an antiviral compound, gemcitabine, that also proved useful for treating cancer. . . . As in Pfizer, we held the patentee's subsequent claims to methods of using gemcitabine to treat cancer invalid for double pateting, looking to the disclosure of anticancer utility in the first patent's specification.
In summary, according to the court, Pfizer and Sun "address the situation in which an earlier patent claims a compound, disclosing the utility of that compound in the specification, and a later patent claims a method of using that compound for a particular use described in the specification of the earlier patent. Furthermore, in each of those cases, the claims held to be patentably indistinct had in common the same compound or composition."
The court distinguished this case:
Rather than a composition and a previously disclosed use, the claims at issue recite two separate and distinct chemical compounds: the '775 Intermediate and pemetrexed, differing from each other in four respects. That alone suffices to undermine Teva's argument regarding the '775 Intermediate, for the asserted claims of the '932 patent do not recite a use of the same compound, but a different compound altogether.
Finally, the Federal Circuit corrected an error made by the district court. The district court disregarded Lilly's objective evidence of nonobviousness, holding that "secondary considerations are not relevant to the analysis of invalidity for obviousness-type double patenting." The Federal Circuit took the opportunity to reiterate its recent statement from In re Cyclobenzaprine: a fact-finder "must withhold judgment on an obviousness challenge until it has considered all relevant evidence, including that relating to the objective considerations."